-
Structure and Characterization of KSc(BH4)4

R. Cerny, D.B. Ravnsbæk, G. Severa, Y. Filinchuk, V. D'Anna, H. Hagemann, D. Haase, J. Skibsted, C.M. Jensen and T.R. Jensen
Journal of Physical Chemistry C, 114 (45) (2010), p19540-19549


DOI:10.1021/jp106280v | unige:14680 | Abstract | Article HTML | Article PDF
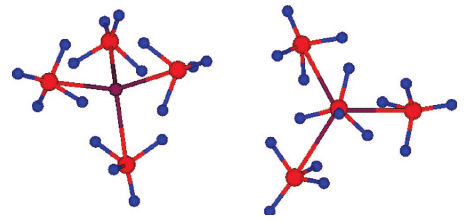
A new potassium scandium borohydride, KSc(BH4)4, is presented and characterized by a combination of in situ synchrotron radiation powder X-ray diffraction, thermal analysis, and vibrational and NMR spectroscopy. The title compound, KSc(BH4)4, forms at ambient conditions in ball milled mixtures of potassium borohydride and ScCl3 together with a new ternary chloride K3ScCl6, which is also structurally characterized. This indicates that the formation of KSc(BH4)4 differs from a simple metathesis reaction, and the highest scandium borohydride yield (~31 mol %) can be obtained with a reactant ratio KBH4:ScCl3 of 2:1. KSc(BH4)4 crystallizes in the orthorhombic crystal system, a = 11.856(5), b = 7.800(3), c = 10.126(6) Ã
, V = 936.4(8) Ã
3 at RT, with the space group symmetry Pnma. KSc(BH4)4 has a BaSO4 type structure where the BH4 tetrahedra take the oxygen positions. Regarding the packing of cations, K+, and complex anions, [Sc(BH4)4]â, the structure of KSc(BH4)4 can be seen as a distorted variant of orthorhombic neptunium, Np, metal. Thermal expansion of KSc(BH4)4 in the temperature range RT to 405 K is anisotropic, and the lattice parameter b shows strong nonlinearity upon approaching the melting temperature. The vibrational and NMR spectra are consistent with the structural model, and previous investigations of the related compounds ASc(BH4)4 with A = Li, Na. KSc(BH4)4 is stable from RT up to ~405 K, where the compound melts and then releases hydrogen in two rapid steps approximately at 460â500 K and 510â590 K. The hydrogen release involves the formation of KBH4, which reacts with K3ScCl6 and forms a solid solution, K(BH4)1âxClx. The ternary potassium scandium chloride K3ScCl6 observed in all samples has a monoclinic structure at room temperature, P21/a, a = 12.729(3), b = 7.367(2), c = 12.825(3) Ã
, β = 109.22(2)°, V = 1135.6(4) Ã
3, which is isostructural to K3MoCl6. The monoclinic polymorph transforms to cubic at 635 K, a = 10.694 Ã
(based on diffraction data measured at 769 K), which is isostructural to the high temperature phase of K3YCl6.